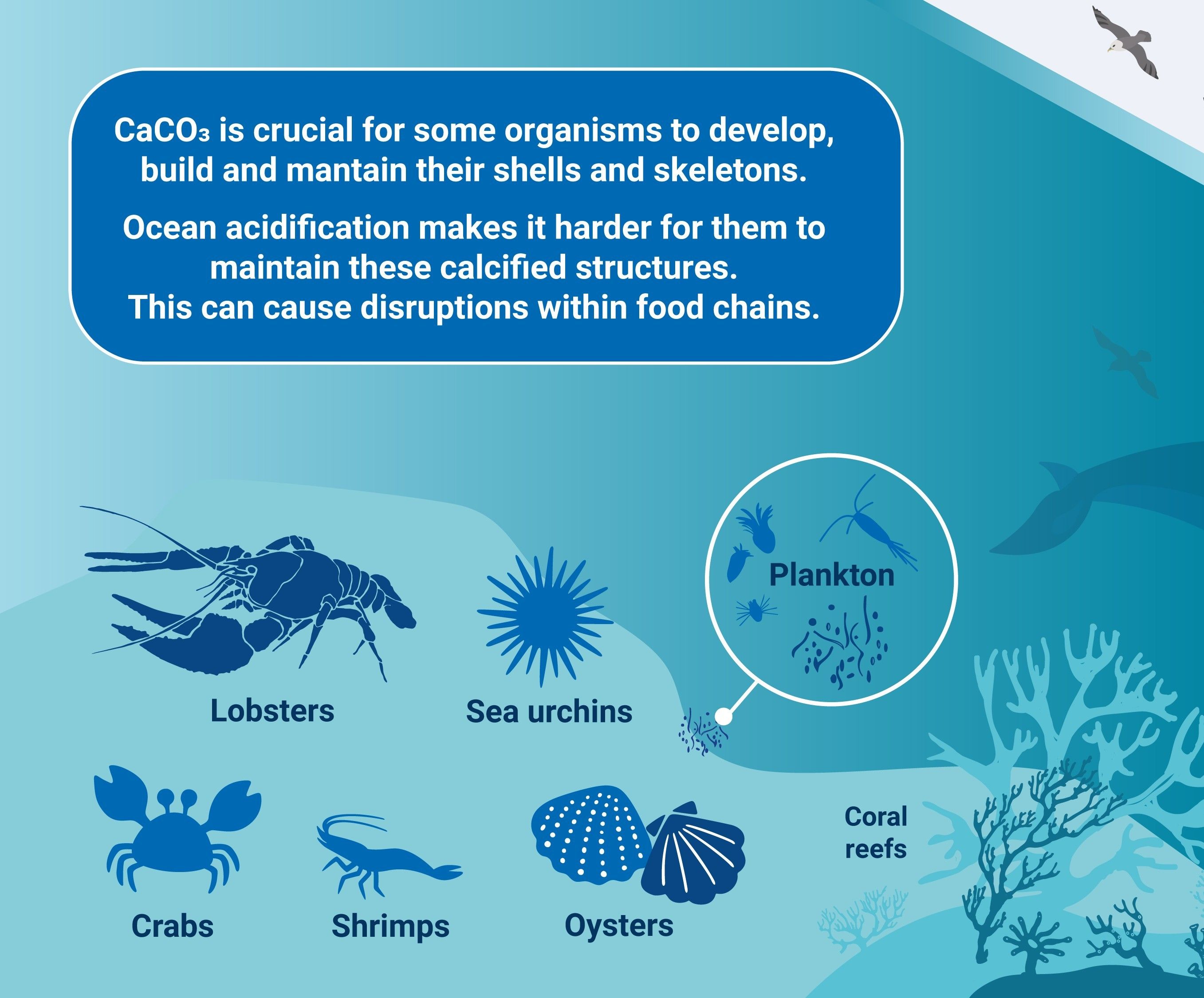
Higher amounts of carbon are now being absorbed by oceans and estuaries, increasing the acidity of these waters (e.g., NOAA Ocean Acidification Program). Hundreds of peer-reviewed research articles detail how the complex chemistry of calcium carbonate in saltwater is changing in ways that affect the skeletal development and other morphological and physiological attributes of species of invertebrates (e.g. corals and mollusks) and fishes (e.g., Mollica, 2018). Many estuarine organisms of the IRL are adapted to differing hydrological and chemical features than adjacent ocean systems. Though there can be much exchange of some species between oceans and estuaries, the potential types and effects of acidification will clearly vary.
In 8 of 10 Florida estuary systems, analysis of >80,000 measurements from 1980 to 2008 showed significant trends in pH decrease (acidification). Decreases were 2.0–3.4 times slower in Tampa Bay than in ocean waters offshore, possibly mitigated by the carbonate shelf platform’s contributions to buffering capacity (Robbins and Lisle, 2018). Tampa Bay pH conditions have been shown to be increasing in some areas with the recent expansions of seagrass habitat (Sherwood et al. 2016).